Decipher Prostate RP
Test Report Overview
We want you to feel confident in every treatment decision you make using Decipher Prostate for your patients with prostate cancer, beginning with a clear understanding of how to interpret the key components of the RP test report.

Page 1 contains the Decipher Score, risk estimates, and an interpretation of the score.

1. Dynamic Report
The report is tailored to the clinical presentation of this patient
2. Decipher Score
- Reflects genomic risk of metastasis
- Determined by tumor biology alone, independent of clinical & pathological factors (e.g., Gleason, PSA)
- 22 genes, 7 biological pathways
- Continuous genomic risk score classified as low, intermediate or high
3. Interpretation
Summary based on this patient’s genomic risk & relevant clinical findings
4. Risk Estimates
- Calibrated to outcomes of patients after surgery with undetectable, persistently-elevated, or rising PSA
- Metastasis at 5 & 10 years
- Prostate Cancer Mortality at 15 years
Page 2 contains case analysis, interpretation, and clinical findings.
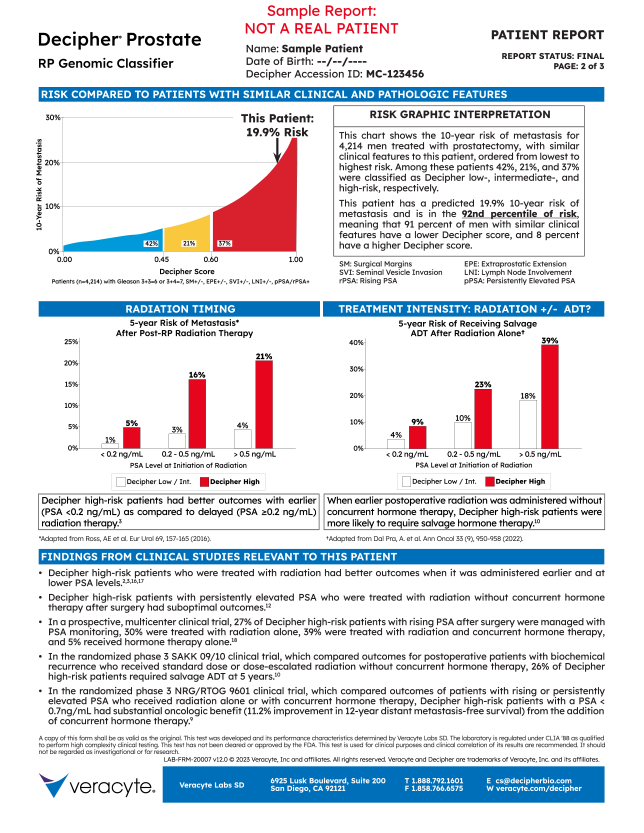
5. Interpretation
- Explains risk comparison graphic
- Provides this patient’s percentile rank in the tested population
6. Risk Comparisons
- This patient’s 10-year risk of metastasis with respect to 3,013 other patients with Gleason 4+3=7 disease & rising PSA
- The distribution of Decipher risk within the 3,013 patients is:
- 19% Decipher Low
- 16% Decipher Int.
- 65% Decipher High
7. Treatment Graphics
- Treatment Timing (Left)
- Risk of metastasis after post-RP radiation stratified by Decipher risk & PSA level at time of treatment
- Treatment Intensity (Right)
- Risk of receiving salvage ADT after radiation alone stratified by Decipher risk & PSA level at time of treatment
8. Clinical Findings
Clinical study results relevant to this patient
Page 3 contains a test description, intended use info, confidence intervals, and references.
9. Test Description
- Description of:
- Decipher testing platform technology
- Risk estimates
- Cut-points separating Decipher low, intermediate & high
10. Intended Use
Decipher Prostate RP is intended for use in post-RP specimens from localized prostate cancer patients with undetectable, persistent, or rising PSA
11. Confidence Intervals
The 95% confidence intervals for each risk estimate (on page 1)
12. References
For each of the clinical studies cited in the report